Accelerated in vitro preclinical evaluation of viral vectors for retinal gene therapy development
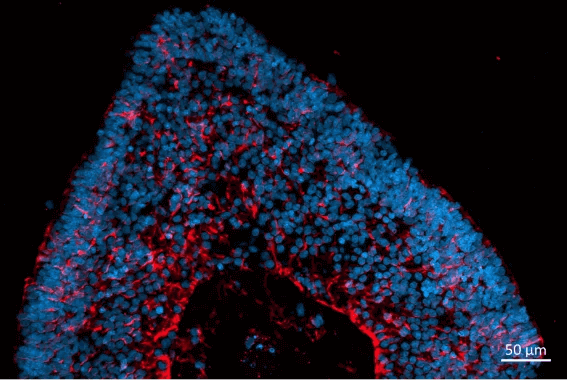
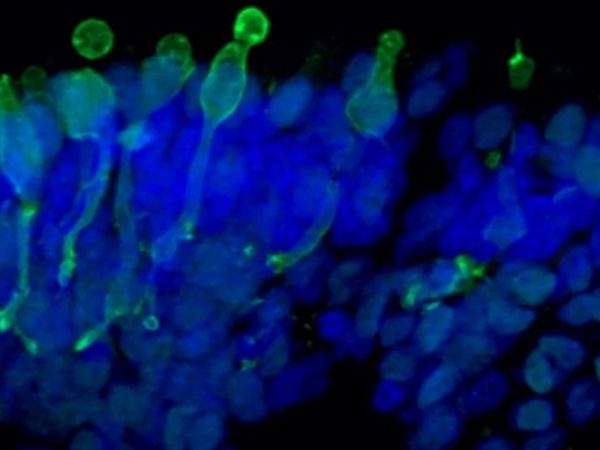
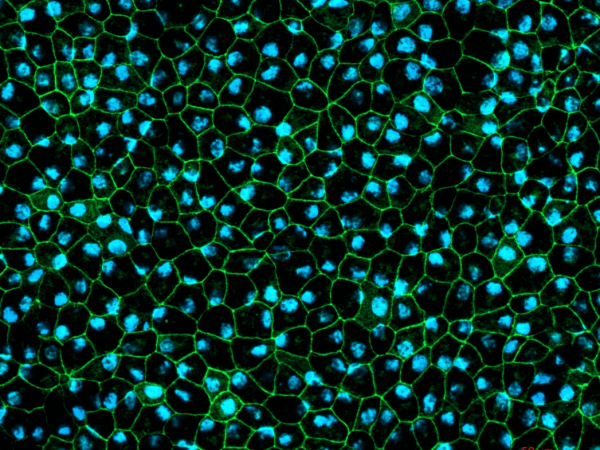
Newcells retinal organoids and RPE models are valuable tools for in vitro preclinical selection of new retinal gene therapy vectors. They allow rapid in vitro evaluation of the best combination of vector capsid, promoter, and transgene. They also enable initial safety and efficacy testing to be carried out using the exact same promoter or gene as those to be used in clinical trials. Our retinal models have been validated for their use in screening and selection of optimal adeno-associated viral (AAV) vectors with increased photoreceptor tropism for retinal gene therapy.
Service outputs
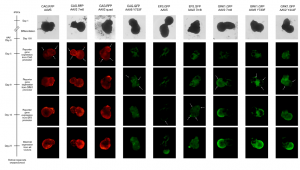
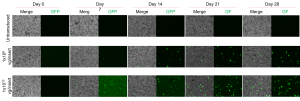
- Evaluation of AAV vectors transduction efficiency in iPSC-derived retinal organoids or RPE cells
- Determination of AAV vector cell tropism and transduction of photoreceptor-like cells in retinal organoids
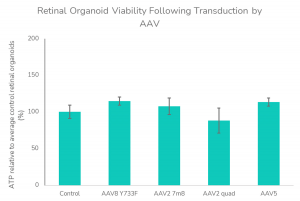
- Cell viability assessment post-transduction